By Prof. Dr Lieven Annemans, expert trainer of the courses
Health Economics for Non-Health-Economists, Basics of Health Economics, and Critical New HTA Developments in Europe: Challenges & Solutions
One of the most underestimated pitfalls in HTA is how treatment effects are presented. The distinction between Relative Risk Reduction (RRR) and Absolute Risk Reduction (ARR) may seem technical, but in practice it can determine whether a product is perceived as valuable or not.
📊 Same data, different narrative
Consider a treatment that reduces event rates from 10% to 8% in comparison with the Standard of Care (SoC):
- Relative Risk Reduction (RRR): (10% - 8%) / 10% = 20%
- Absolute Risk Reduction (ARR): 10% - 8% = 2%
Below is a simple way to visualise this:
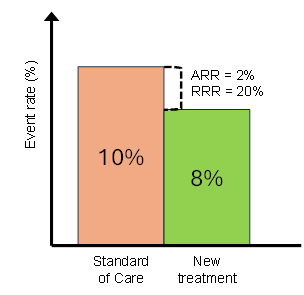
What stands out immediately:
The relative drop looks large, but the absolute difference is small.
Both are correct. Yet they lead to very different interpretations.
⚖️ Why payers care about ARR
HTA bodies focus on ARR because it feeds into the core elements of decision-making:
- Clinical relevance: ARR reflects the real number of patients benefiting
- Cost-effectiveness: ARR determines the incremental health benefit used in QALYs and ICERs
⚠️ The SoC risk problem
The gap between RRR and ARR becomes critical when the risk with SoC is low.
A strong RRR can still translate into minimal absolute benefit. This is where some submissions lose credibility with payers.
In contrast, higher-risk populations increase ARR, which improves both:
- perceived clinical value
- cost-effectiveness outcomes
This explains – together with the aim to reduce budget impact – why HTA bodies often push for more targeted populations.
📝 Implications for evidence generation
- Consider expected ARR before selecting trial populations
- Always present absolute outcomes clearly
- Align messaging with payer logic, not just clinical storytelling
- Present the absolute benefits also on population level (how many events avoided in total)
📌 The bottom line
RRR often makes the effect look impressive.
ARR extrapolated to population level determines whether it matters.
And in HTA, what matters is what gets reimbursed.
Continue your learning from Lieven
If you’d like to learn more from Lieven, CELforPharma also offers a 1-day, hands-on course where you'll learn:
- The implications of the EU HTA Regulation and Joint Clinical Assessments
- How national HTA processes are adapting – and what this means for your strategy
- Real-world challenges companies face in aligning evidence, value, and access
- Practical approaches to evidence generation and strategic preparation in a changing HTA environment
Don’t miss the latest insights from our expert faculty
Subscribe to our newsletter to:
- Stay on top of the latest expert insights
- Receive invitations to upcoming educational webinars
- Get updates on our courses and training programmes
